| |
Hands-On, Minds-On Meteorology
Description
| Programming | Operation
| Evaporation
Description
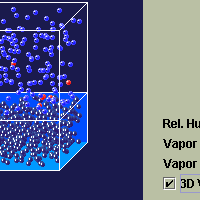
The Evaporation application is designed to the allow the students to
see the process of evaporation and condensation between the ocean and
the atmosphere.
|
click for whole shot |
Objectives
The primary objective of the Evaporation program is to give the students
a sense of the never-ending equilibrium condition of water vapor. The
speed at which humidify is transferred from one system to the other
is graphically displayed.
Programming
Theory
The Evaporation program allows students to view a microscopic volume
that is right at the air-ocean interface. The bottom part of the volume
is in the ocean, while the upper part is in the air. In the air, the
water vapor and liquid water molecules are shown, but unlike other
programs where this is the case, dry air molecules are never shown.
Red-colored particles are shown in this program, but they represent
molecules that have just changed from the ocean to the air or vice-versa.
Also, water in the ocean is represented by dark blue particles.
Students begin the experiment by choosing an ocean temperature and
an air temperature. Upon clicking the start button, ocean water molecules
will begin to move into the air, turning red for a few time steps,
and then turning blue -- the normal color for water vapor in this
program. Some of these vapor molecules may condense into liquid water,
some may return to the ocean, turning red again, then the dark blue
color that represents ocean water. Ultimately, the Relative humidity
will escalate into the 90-100% range, but how quickly depends on the
initial settings of the ocean and air.
Assumptions
See ParcelSet*
Graphics and Visualization
Ocean water is differentiated from other forms of water by making
it a little darker (dkbluesm.gif). Red particles in Evaporation
do not represent dry air molecules, they represent molecules that
have recently changed phase.
Once condensation has occurred, it is often difficult to determine
how many water molecules are included in the clump, so when a condensed
liquid droplet has accumulated 4 or more water molecules, a small
number is placed above it.
Also, at the moment of condensation, a small quick explosion-looking
image is shown and quickly disappears. These flashes represent the
release of latent heat for that individual condensation occurrence.
Also See Condensation
Condensation Process
See Condensation and Water
Collisions.
Changing a parcel form ocean to atmosphere and back
The calculations made to determine this process are arbitrary.
If a liquid water molecule in the ocean comes into contact with
the ocean-atmosphere interface at a speed greater than 800 m/s,
it is allowed to pass into the atmosphere. If a water molecule (liquid
or vapor) in the atmosphere comes into contact with the ocean-atmosphere
interface at a speed less than 250 m/s, it is allowed to pass into
the ocean. If a condensed water particle strikes the interface with
a slow enough speed, it will give one water vapor molecule to the
ocean, not the entire particle.
For ease of viewing, parcels entering the atmosphere from the ocean
always do so on the right side of the cube, and atmospheric parcels
entering the ocean do so on the left side of the cube, regardless
of where they hit the interface.
Equations
See ParcelSet*
Other
See ParcelSet*
Operation
Running the Program
- Click the link for Evap.
- Change the ocean temperature by adjusting the Ocean Temperature
scrollbar.
- Change the atmospheric temperature by adjusting the Atmospheric
Temperature scrollbar.
- Click the 'Stop' button to pause the simulation. The button's
label will change to 'Start'.
- Click the 'Start' button to resume the simulation. The button's
label will change to 'Stop'.
- Click the 'Reset' button to reset the simulation. This will remove
all water vapor from the atmosphere. Click 'Start' to then begin
a new simulation.
- Uncheck the '3D View' checkbox to view the volume head-on. This
view helps to distinguish the red particles that represent both
new atmosphere and new ocean water molecules.
Extra Knowledge
Any other information
 |
Department of Atmospheric Sciences
University of Illinois at Urbana Champaign
Created by Dan Bramer: Last Modified 07/27/2004
send questions/comments to bramer@atmos.uiuc.edu
|
|







